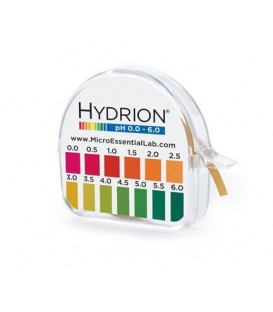
pH Litmus Paper Test Strip Roll 3.0 - 5.5 (325)
pH Litmus paper 3.0 - 5.5 is a short-range general use pH Hydrion Litmus paper for measuring the pH of acidic solution including woman's health. Each roll is 6mm (1/4in) wide x 4.5 metres (15ft) and will give the user approximately 100 tests. Comes complete with colour chart and directions.
Volume discounts
| Quantity | Discount | You Save |
|---|---|---|
| 5 | 5% | Up to $4.99 |
| 10 | 10% | Up to $19.95 |
pH Litmus Paper Test Strip Roll 3.0 - 5.5 (325)
This short-range Hydrion Brilliant pH Paper provides excellent color separation in the acid range and offers clear bright single color matches at every .5 interval from pH 3.0-5.5.
This Hydrion pH Paper may be used in diverse pH applications including:
Woman's Health, Classroom Demonstrations, Hospital Laboratories, General Laboratories, Industrial Testing, Food Service, Safety Testing, Emergency Testing, Agricultural Testing In Vitro Testing.
Hydrion pH Litmus Paper Test Strip Roll pH 3.0 - 5.5 is the most economical option to purchase Hydrion pH Litmus Paper.
It has excellent colour separations to suit a wide variety of measurement requirements in the acid range (below pH 7).
The colour chart has pH matches at...

With a measuring range of 3.0 - 5.5 and precise pH matches at every 0.5 pH intervals makes the Hydrion pH Litmus Paper Test Strip Roll the preferred choice of our customers.
- Each roll will give the user approximately 100 tests
- 6mm (1/4in) wide x 4.5 metres (15ft)
- A direct reading pH paper with the widest range available in a single paper
- A simple, single color match for all pH values
- Distinct and easily matched color separations
- Accurate, reproducible readings with an immediate response
As a safety precaution, an unknown solution should be measured for its pH value. If it is found to be too acidic, the solution may be dangerous to handle, or may have special disposal requirements.
Please note: Our range of pH testing equipment are genuine Hydrion pharmaceutical grade quality and not cheap imports. Hence your readings will be instant and accurate as is required in laboratory conditions.
Directions for use
To use this product simply tear off a small strip (50mm - 2½") of pH paper and dip into test solution.
Compare the instantaneous colour change with the matching pH colour chart.
Related video
Frequently Asked Questions
Approximately how many tests per roll is there?
There is approximately 100 tests per roll depending on the length of paper you tear off each time. For instance, if you used a strip of paper at 25mm or 1 inch, you'll get close to 180 tests!
Can I use my pH Litmus Test Paper 3.0 - 5.5 to measure the pH of urine & saliva?
No we do not suggest using pH 3.0-5.5 for measuring urine and saliva pH.
For accuracy in testing urine and saliva pH, we suggest using finer calibrated testing equipment such as...
These two measuring products have pH measurement intervals in the 0.2 making them the better option for accurate urine & saliva pH readings.
Can I use my pH Litmus Test Paper to measure the pH of water?
<pWhat is a buffered solution?
A buffered solution is a solution with adequate dissolved solids to resist pH changes. For example, urine and saliva are considered buffered in their natural state.
However, rain water, tap water etc. are considered to be dilute, weakly buffered solutions.
For testing weakly buffered solutions, we recommend our water pH testing kits.
What do the results indicate?
If you have high pH levels – you have low acid levels.
If you have low pH levels – you have high acid levels.
This reading in turn can be used to adjust your diet and mineral intake so as to bring cellular terrain back into balance.
Related articles
| pH Range | 0.0-0.6 |
| Measurement Increments | 0.5 pH units |
| Number of Tests | 100 |
| Used to Test | Buffered solutions in acidic range |
| Country of Origin | Brooklyn USA |
| Material | Hydrion litmus paper roll housed within plastic case. Case has serrated edge to tear off paper. |
| Colour Chart | Yes |
| Manufacturer | Micro Essential Laboratory |